 
nitrogen atom has a stronger effective nuclear charge than lithium due to its greater number of protons in the nucleus holding the electrons tighter.Why can we relate classical physics to quantum mechanics when it comes to subatomic activity? For an atom or an ion with only a single electron, we can calculate the potential energy of an electron by considering only the electrostatic attraction between the positively charged nucleus and the negatively charged electron. 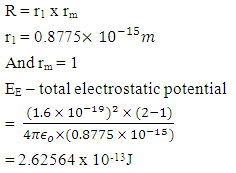
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
